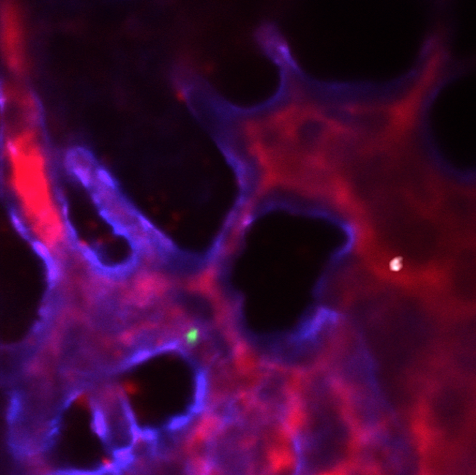
 Above, the CCR2- blood stem cell (green) is positioned closer to bone (blue) compared to the rare CCR2+ blood stem cells (white). Generally, cells closer to the bone are more likely to be "quiescent," or inactive, matching the observation that the CCR2+ HSC population proliferate after a heart attack.
Above, the CCR2- blood stem cell (green) is positioned closer to bone (blue) compared to the rare CCR2+ blood stem cells (white). Generally, cells closer to the bone are more likely to be "quiescent," or inactive, matching the observation that the CCR2+ HSC population proliferate after a heart attack.
Harvard Stem Cell Institute (HSCI) researchers, and scientists at Massachusetts General Hospital (MGH) have discovered a rare type of blood stem cell that controls the production of “emergency” white blood cells whose appearance after a first heart attack, while vital, may lead to serious and sometimes fatal health complications, such as a second cardiac event or stroke.
The research, published on line in the journal Cell Stem Cell, could eventually lead to the development of a treatment to regulate the number of white blood cells produced, thereby tempering their impact.
The study builds on work by Matthias Nahrendorf, a researcher at MGH, who in 2012 determined that shortly after a cardiac event, the sympathetic nervous system—responsible for maintaining “status quo” in the body—sends out a distress signal. As a result, blood stem cells and progenitor cells leave their home in the bone marrow, circulate through the body, and increase their production of “emergency” white blood cells.
While the 2012 research provided insight into how excess white blood cells are produced, it revealed only part of the picture; precisely which cells received that signal remained unknown, Nahrendorf said.
Nahrendorf, who since the beginning of his career has been studying the mechanisms of heart attacks and is the current study’s senior author, wanted to pinpoint exactly where in the production line—from stem cells to the various intermediate progenitor cells to white bloods cells—that nervous system signal gets sent.
“It could have been that a subset of blood stem cells were dormant and not alerted until the heart attack, or it could happen more downstream in the progenitor cells,” he said.
To find out, he collaborated with HSCI imaging expert Ralph Weissleder and to HSCI co-director David Scadden, a blood stem cell expert.
Together, with the paper’s first author, Partha Dutta, and Filip Swirski of MGH, the research team found a rare subset of blood stem cells — those capable of activating the CCR2 gene — acted as gatekeepers to emergency white blood cell production.
Of all blood stem cells, only 15%, called CCR2+, are capable of activating the CCR2 gene. They generally remain dormant in the bone marrow. During a heart attack, however, the nervous system signals the CCR2+ cells to leave the bone marrow and mass-produce progenitor cells that quickly give rise to the emergency white blood cells.
“You need these white blood cells to go to the heart and remove the dead tissue there, but if you have too many it gives you heart failure,” Nahrendorf said. When too many white blood cells are present, the excess can turn and attack the heart lining and other vital organs, destroying to the very tissues they were meant to protect.
After a heart attack, physicians often monitor the number of white blood cells in order to understand the likelihood of the patient’s survival—patients with high white blood cell counts (more than 7,000 cells per cubed millimeter of blood) are nearly twice as likely to experience another heart attack or other potentially life-threatening complications, such as a stroke or irregular heart beat.
Additionally, the excess white blood cells can turn away from the blockage site, travel to different parts of the circulatory system, and create new plaques.
While these white blood cells only live a matter of days, the damage they inflict can be lasting; one out of every two survivors will have a second heart attack or further coronary complications within a year of their first heart attack.
However, Nahrendorf said, simply shutting off the CRR2 gene isn’t the answer; CCR2 helps direct cells from the bone marrow and spleen to the blockage site in the heart.
When the researchers studied heart attacks in mice that don’t have a working CCR2 gene, they found white blood cells made by other blood stem cell types couldn’t migrate out of the bone marrow.
“We believe there is a sweet spot,” Nahrendorf said. The cells needs to make enough white blood cells to repair the heart and stave off foreign bacteria, viruses, and parasites, especially if a patient is post-surgery and vulnerable to infection, but not too many that they cause more harm.
The researchers hope that understanding the basic biology of how this system works will eventually allow them to find ways to target those specific stem cells and “dampen” their effects without completely shutting them off.
